The Future of Manufacturing Risk Intelligence
FMEAi transforms how medical device teams perform Failure Mode & Effects Analysis. Stop guessing failure modes from blank spreadsheets. Start predicting them with AI.
Trusted by medical device teams. Validated by peer-reviewed research.
92% Accuracy
AI Prediction Rate
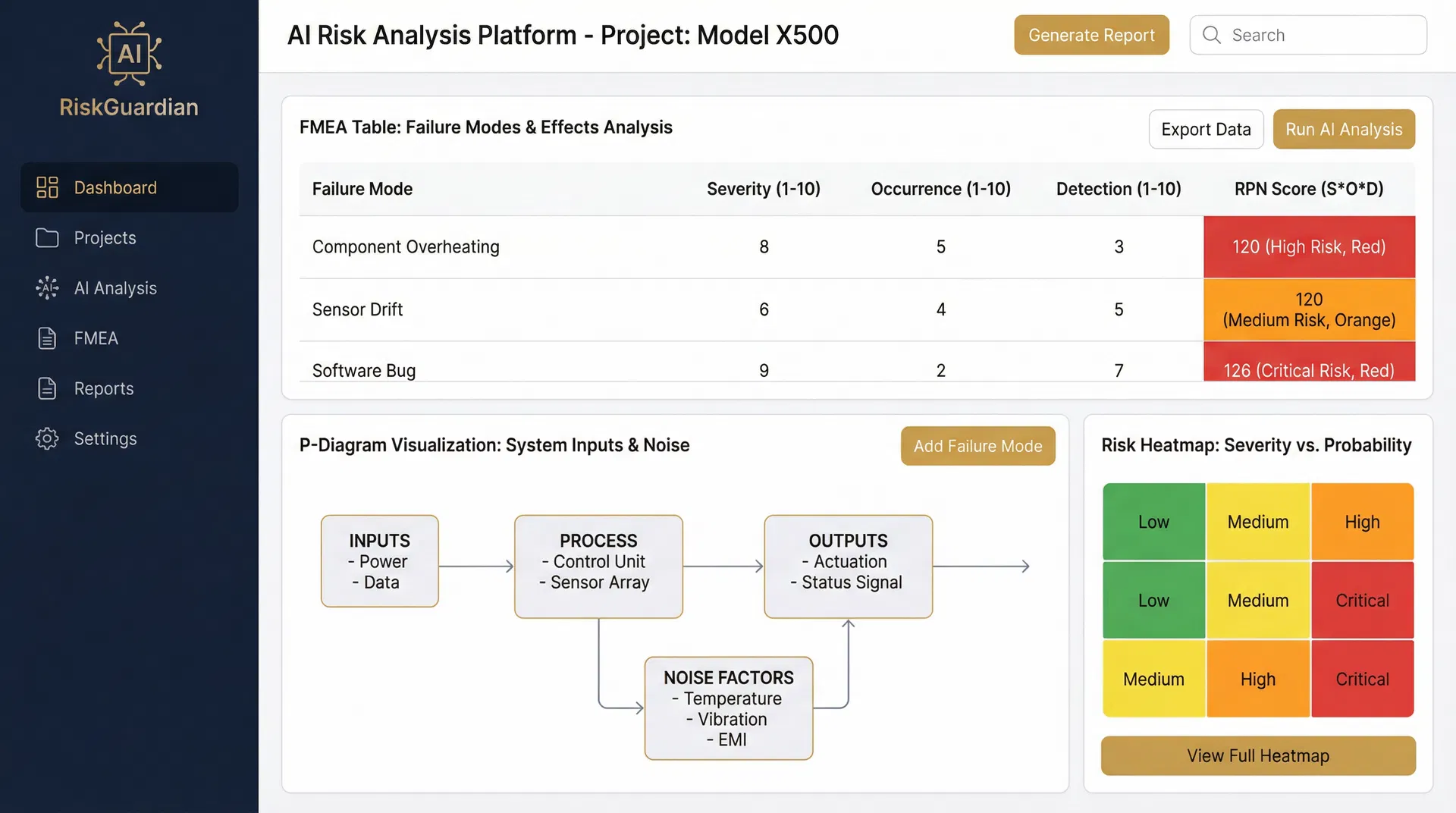
The $100B Manufacturing Risk Crisis
60% of manufacturers struggle with design risk management. Engineers start from blank spreadsheets, manually guessing failure modes with zero AI assistance. The result? Incomplete analysis, missed risks, and costly recalls.
"It's not very interactive... it's not very easy and intuitive to understand what it is."
— Senior Design Assurance Engineer, Fortune 500 Medical Device Company
9 Integrated Features. All AI-Powered.
Every feature is designed to eliminate manual guesswork and embed regulatory intelligence directly into your risk analysis workflow.
FMEA Wizard
8-step guided assessment per IEEE 1633 & ISO 14971. Upload design docs and let AI guide you through a standards-based risk analysis.
AI Failure Mode Generation
LLM analyzes your design inputs to suggest failure modes, causes, effects, S/O/D ratings, and RPN scores — no more blank spreadsheets.
P-Diagram Generator
Auto-derives Parameter Diagrams with inputs, outputs, noise factors, and control factors. Click any unintended output to generate FMEA items.
Dynamic FMEA Table
Spreadsheet-style editor with inline editing, sorting, filtering, and real-time RPN calculation. Export to Excel and CSV with full traceability.
Gap Analysis Dashboard
Import existing FMEAs, compare against AI-derived failure modes, and highlight missing mitigations with occurrence underestimation flagging.
Coverage Tracker
Visual map ensuring your entire design spectrum is analyzed. Know exactly when your analysis is complete with a clear definition of done.
Real-Time FDA Data
Integrated with 4 openFDA APIs — MAUDE Adverse Events, Device Recalls, Enforcement Reports, and 510(k) Premarket data for evidence grounding.
AI Chat Assistant
Contextual assistant providing standards guidance, mitigation tips, and risk analysis support throughout your entire FMEA workflow.
Multi-Project Management
Full project CRUD with regulatory context, device metadata, and team collaboration. Manage your entire risk portfolio in one place.
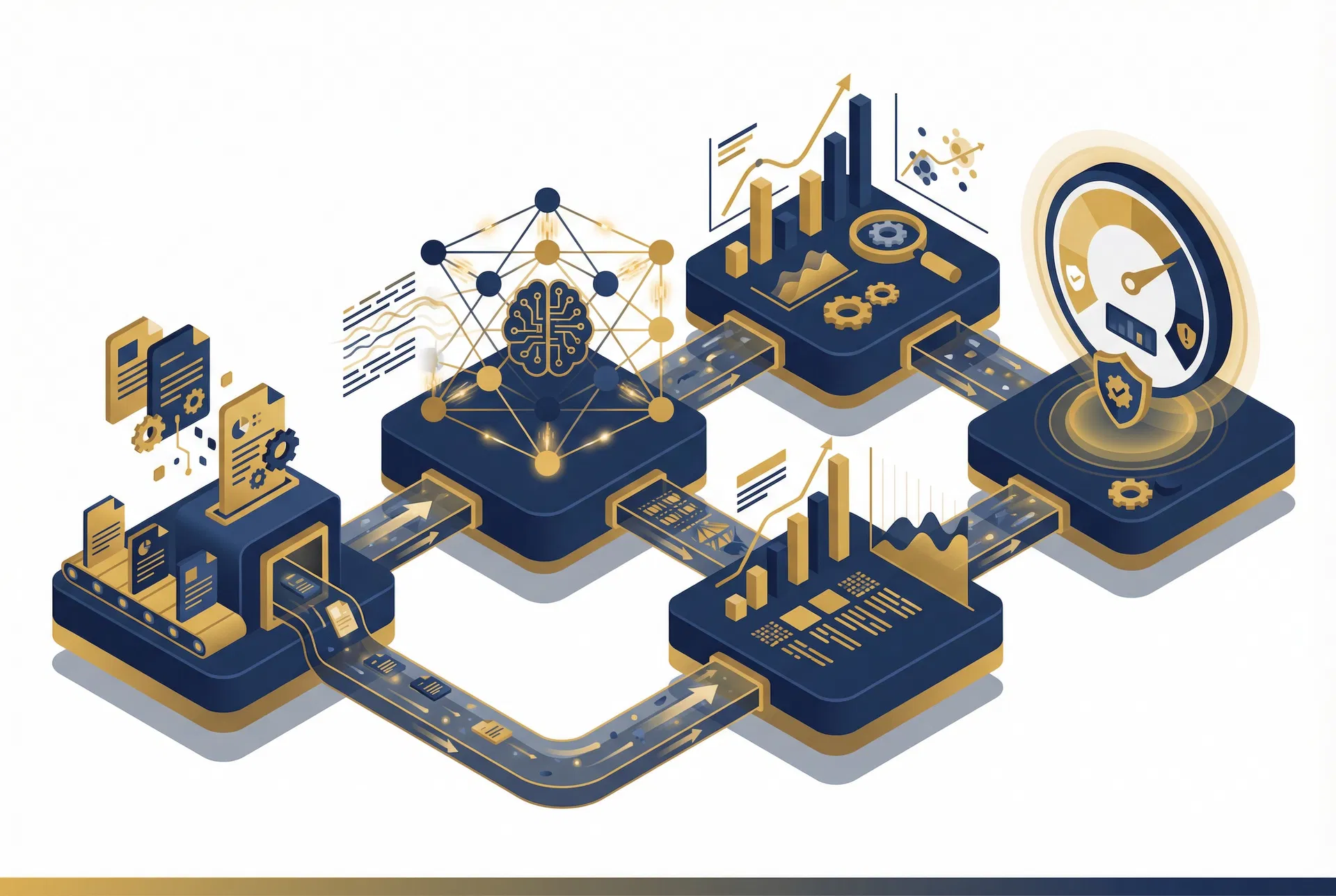
From Design Docs to Risk Intelligence in Minutes
Data Collection
Upload design docs, requirements, architecture specs, and existing FMEAs. FMEAi ingests everything.
NLP Processing
Our LLM auto-tags failure modes via structured JSON schema, extracting risk patterns from your design inputs.
Predictive Analytics
Risk forecasting calibrated to ISO 14971 Severity/Occurrence/Detection frameworks with 92% accuracy.
Dynamic Risk Scoring
RPN scores recalibrated using real-world clinical and field data from FDA databases. Patent-pending technology.
P-Diagram to FMEA: The Connection No Other Tool Makes
Click any unintended output on a P-diagram and FMEAi instantly generates a complete FMEA line item — with failure mode, cause, effect, S/O/D ratings, and FDA evidence grounding. No other tool connects architecture diagrams directly to risk analysis with real-time regulatory data.
"If that connection with existing architecture and design is made, it immediately brings out a usefulness for the tool."
— Senior Design Assurance Engineer, Customer Discovery Session
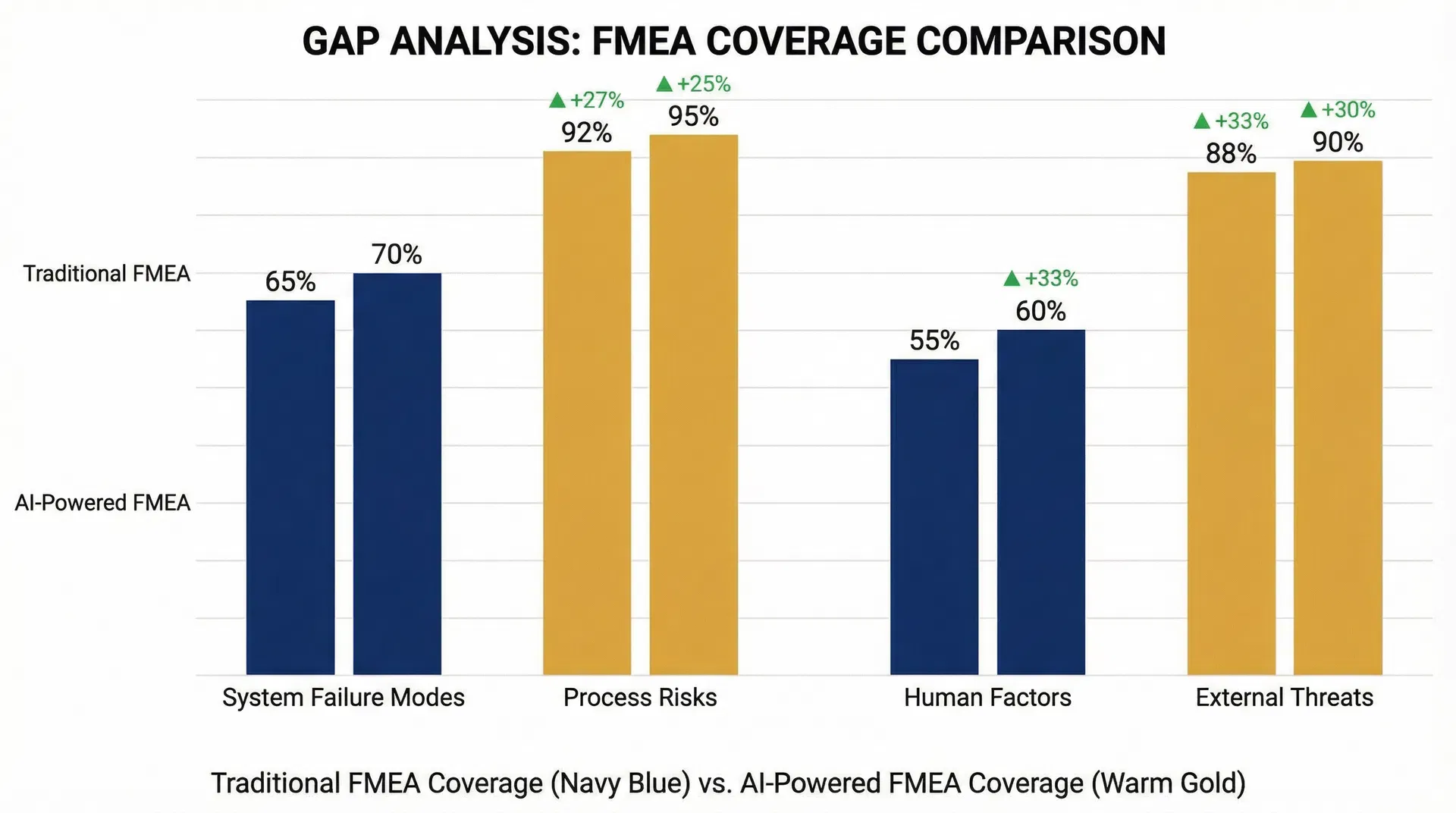
AI-First vs. Legacy Spreadsheet Tools
| Feature | Traditional FMEA | FMEAi |
|---|---|---|
| Data Entry | Manual, error-prone | AI-automated from design inputs |
| Risk Scoring | Static, subjective | Dynamic, data-calibrated |
| Standards Compliance | Generic templates | Embedded regulatory intelligence |
| Architecture Link | Isolated spreadsheets | Integrated P-diagrams & design |
| Gap Detection | None | AI-powered field data comparison |
| Prediction | Reactive | 92% accurate proactive forecasting |

Built-In Standards Compliance
FMEAi embeds regulatory intelligence directly into your workflow. Every analysis is automatically aligned with the standards that matter most.
ISO 14971
Risk Management for Medical Devices
IEC 62304
Medical Device Software Lifecycle
IEEE 1633
Software Reliability Practices
AAMI TIR57
Medical Device Security
FDA 21 CFR 820
Quality System Regulation
FDA AI/ML
AI-Enabled Device Software Guidance
Backed by Peer-Reviewed Science
FMEAi's methodology is validated by published research with human-in-the-loop expert validation.
AI-Powered FMEA for Cardiovascular Devices
Dhole, 2025
AI-enhanced FMEA on implantable cardioverter defibrillators uncovered 2 legacy low-priority failure modes with remarkably high real-world incident rates. Traditional RPN: 128 → AI-Calibrated: 96.
Integration of LLMs for Faster & More Accurate Risk Analysis
El Hassani et al., 2025
Demonstrates how Large Language Models can be integrated into FMEA workflows to significantly reduce analysis time while improving accuracy and coverage of failure mode identification.
Proven Results, Real Customers
Successful Pilot Deployments
Beta Users Report 50%+ Time Savings
AI Prediction Accuracy (Patent-Pending)
Enterprise Projects by HighCloud
Plans That Scale With Your Team
Start with Starter, grow to Professional. Every plan includes AI-powered risk intelligence and standards compliance.